| SpectraBase Compound ID | 5I0siqJN7U4 |
|---|---|
| InChI | InChI=1S/C74H116O6/c1-4-7-10-13-16-19-22-25-28-31-33-35-36-37-38-40-41-43-46-49-52-55-58-61-64-67-73(76)79-70-71(69-78-72(75)66-63-60-57-54-51-48-45-30-27-24-21-18-15-12-9-6-3)80-74(77)68-65-62-59-56-53-50-47-44-42-39-34-32-29-26-23-20-17-14-11-8-5-2/h7-8,10-11,16-17,19-20,25-26,28-30,33-35,37-39,41,43-45,47,49,52-53,56,71H,4-6,9,12-15,18,21-24,27,31-32,36,40,42,46,48,50-51,54-55,57-70H2,1-3H3/b10-7-,11-8-,19-16-,20-17-,28-25-,29-26-,35-33-,38-37-,39-34-,43-41-,45-30-,47-44-,52-49-,56-53- |
| InChIKey | QEHVSQYTNZCQBK-JFDHSVHQNA-N |
| Mol Weight | 1101.7 g/mol |
| Molecular Formula | C74H116O6 |
| Exact Mass | 1100.877191 g/mol |
Mass Spectrum (LC)
View the Full Spectrum for FREE!
The full spectrum can only be viewed using a FREE account.
| SpectraBase Spectrum ID | 1PXBhWP5g6 |
|---|---|
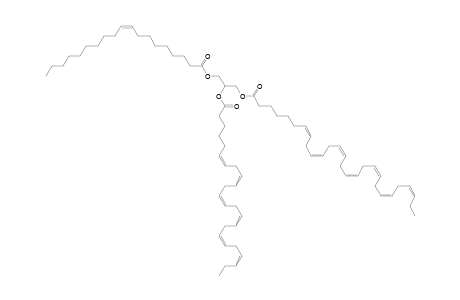
| Name | TG 19:1_24:6_28:7 |
| Classification | Glycerolipids [GL] |
| Comments | Triacylglyceride |
| Copyright | Copyright © 2025 John Wiley & Sons, Inc. All Rights Reserved. |
| Exact Mass | 1100.877191449 u |
| Formula | C74H116O6 |
| InChI | InChI=1S/C74H116O6/c1-4-7-10-13-16-19-22-25-28-31-33-35-36-37-38-40-41-43-46-49-52-55-58-61-64-67-73(76)79-70-71(69-78-72(75)66-63-60-57-54-51-48-45-30-27-24-21-18-15-12-9-6-3)80-74(77)68-65-62-59-56-53-50-47-44-42-39-34-32-29-26-23-20-17-14-11-8-5-2/h7-8,10-11,16-17,19-20,25-26,28-30,33-35,37-39,41,43-45,47,49,52-53,56,71H,4-6,9,12-15,18,21-24,27,31-32,36,40,42,46,48,50-51,54-55,57-70H2,1-3H3/b10-7-,11-8-,19-16-,20-17-,28-25-,29-26-,35-33-,38-37-,39-34-,43-41-,45-30-,47-44-,52-49-,56-53- |
| InChIKey | QEHVSQYTNZCQBK-JFDHSVHQNA-N |
| Ion Polarity | P |
| Literature Reference | Tsugawa, H.; Ikeda, K.; Takahashi, M.; Satoh, A.; Mori, Y.; Uchino, H.; Okahashi, N.; Yamada, Y.; Tada, I.; Bonini, P.; Higashi, Y.; Okazaki, Y.; Zhou, Z.; Zhu, Z.-J.; Koelmel, J.; Cajka, T.; Fiehn, O.; Saito, K.; Arita, M.; Arita, M. A Lipidome Atlas in MS-DIAL 4. Nature Biotechnology 2020, 38 (10), 1159–1163. |
| Literature Reference DOI | https://doi.org/10.1038/s41587-020-0531-2 |
| Precursor Ion | [M+NH4]+ |
| SMILES | CCCCCCCCC\C=C/CCCCCCCC(=O)OCC(COC(=O)CCCCC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CC)OC(=O)CCCC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CC |
| Sample Comments | theoretical MS2 created from the information of SCIEX 5600 and 6600 with 45CE +-15CES |